Does COVID-19 Vaccine Accelerate Neurodegenerative Disease?
Neurological Complication Post COVID 19 Vaccines
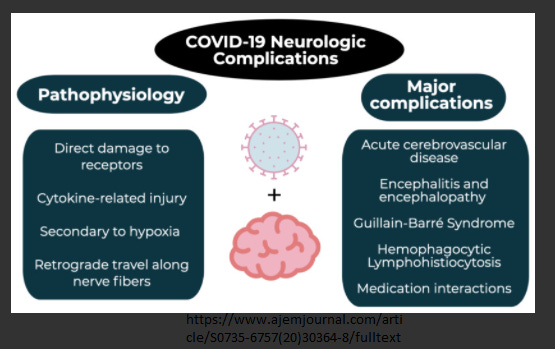
Vaccine is a protection that has been applied since the ancient time. This still on-going COVID-19 pandemic has produced numerous vaccine studies and researches. One of the most important issues is the safety issue. Since this is related to the massive numbers of humans’ lives, it is unethical to weight out the risk of the disease itself to the vaccine adverse effects. Neurological complications after COVID 19 vaccines are not rare. Yellow Card data1 per 22/09/2021 showed some reports for epilepsy, encephalopathy and seizures, Guillain-Barre Syndrome, strokes, Bell’s Palsy, Multiple Sclerosis, Alzheimer’s Disease, hydrocephalus, Parkinson’s Disease, etc. If there is any doubt of the Yellow Card data, some studies have published their data and this article will display some of them.
Yellow Card Data per 22/09/2021 on Tremor, Amnesia, Memory Impairment
Tremor was reported for 1407 cases in Pfizer group, 9697 cases in Astra Zeneca group, and 195 cases in Moderna group. Amnesia was 150 reports in Pfizer group, 384 reports in Astra Zeneca group, and 17 reports in Moderna group. Memory impairment was reported 159 in Pfizer group, 355 cases in Astra Zeneca group, and 17 cases in Moderna group. Parkinsonism was reported 26 in Pfizer group, 195 cases in Astra Zeneca group, and 1 cases in Moderna group. Encephalitis was reported 29 cases in Pfizer group, 57 cases in Astra Zeneca group, and 1 cases in Moderna group. Epilepsy was reported 169 cases in Pfizer group, 375 cases in Astra Zeneca group, and 8 cases in cases Moderna group. All of these numbers were from Yellow Card data.
A View from A Controlled Variable
A nonCOVID-19 unrelated preprint paper about the PD (Parkinson’s Disease) in East London Primary Care2, showed the strongest PD associations were tremor and ′memory complaints′, less than 2 years before PD diagnosis[2]. Associations for both complaints persisted up to 10 years prior to PD diagnosis. Shoulder pain was more common in those who developed PD, emerging 5 to 10 years prior to diagnosis and may be a surrogate marker for rigidity. Epilepsy showed a strongest association with subsequent PD and associations were also found for hypertension and type 2 diabetes, 5 to 10 years before diagnosis. A weak but novel association was observed with prodromal hearing loss and subsequent PD, which appeared up to 10 years prior to diagnosis. From this study we can say that the earliest marker for PD in the Primary Care were tremors and memory complaints, in less than 2 years. Epilepsy was showed 5 to 10 years before diagnosis. The study was published in the early of October 2021. The red lines of this study are tremor related to PD can be seen in less than 2 years before; and epilepsy is detected 5-10 years before the diagnosis.
COVID-19 Vaccine and Epilepsy
Massoud3 et al did survey on people with epilepsy (PwE) who took COVID 19 vaccine i.e. Pfizer BioNTech mRNA vaccine and the Oxford-Astra Zeneca chimpanzee adenovirus-vectored vaccine (ChAdOx1nCoV-19) in Kuwait. Researchers concluded that that the two vaccines under consideration, have a good safety profile and a low risk of epilepsy worsening among a cohort of PwE in Kuwait. The study’s method was a questionnaire to PwE, who visited the epilepsy clinic at Ibn Sina Hospital in Kuwait during the first two working weeks of April 2021. We can conclude that the participants were likely taking the epilepsy medications.
Liu4 et al reported two patients with no known neurological or psychiatric history who presented with encephalopathy and seizures that began after part one of their Moderna COVID-19 vaccine series. Both patients had delirium and non-convulsive status epilepticus in the setting of an extensive negative workup. The duration of symptoms is inconsistent with seizures and altered mental status from acute intoxication or withdrawal from illicit drugs. The team also mentioned that other researchers reported a 68-year-old rural Indian man experiencing non-epileptic seizures four days after receiving his first dose of Covishield vaccine.
Case 1: An 86-year-old woman with a history of diastolic dysfunction, chronic kidney disease stage 3, glaucoma, cataracts, and Type 2 diabetes mellitus was hospitalized 7 days after receiving her first Moderna COVID-19 vaccine administration.
Case 2: A 73-year-old man with a history of Crohn’s, hereditary hemochromatosis, hypertension, and hyperlipidemia was hospitalized 21 days after receiving his first Moderna COVID-19 vaccine. He was recently hospitalized for 2 days following 10 days of staring episodes, restlessness, and cognitive deficits that started 7 days after his Moderna COVID-19 vaccine. There were no new medications except for a prednisone 40mg taper that was started 5 weeks ago for a Crohn’s flare. Prednisone (has anti-inflammatory and immunomodulating properties) was discontinued at the prior 2-day hospitalization and the patient was discharged in improved condition following IV fluid administration and stable admission course, at patient and wife’s request. While at home, he developed hallucinations, worsening confusion and continued to have periods of unresponsiveness. According to the researchers, to suddenly develop primary epilepsy at the age of 73 would therefore be extremely unlikely.
Liu et al concluded that increased inflammatory signals can increase the likelihood of seizures, which is likely why there is an increased prevalence of seizures in intensive care unit COVID-19 patients. Patient 2 had taken anti-inflammatory drug before first admission after the vaccine, The team proposed that the production of the spike protein, from cells translating the mRNA, triggers the same inflammatory cascade as a COVID-19 infection, resulting in these neurotropic effects. They proposed, “This common COVID-19 related CNS complication is caused by a cytokine storm triggered by the virus itself. Severe acute respiratory syndrome coronavirus 2 (SARS-COV-2) has a spike glycoprotein that can bind the angiotensin-converting enzyme 2 (ACE-2) receptor. ACE2 binding and subsequent inhibition has been hypothesized to decrease brain-derived neurotrophic factor (BDNF). This theoretically reduces BDNF's anti-inflammatory action on neurons and its ability to attenuate microglial activation. Increased inflammatory signals can increase the likelihood of seizures, which is likely why there is an increased prevalence of seizures in intensive care unit COVID-19 patients.”
COVID-19 Vaccine and Encephalopathy
Al-Mashdali5 et al reported, a 32-year-old previously healthy man who developed acute confusion, memory disturbances, and auditory hallucination within 24 hours from getting his first dose of the COVID-19 Moderna vaccine. EEG showed features of encephalopathy, CSF investigations were nonspecific, and MRI head did not depict any abnormality. The team discussed that, “Extensive workup for different causes of acute encephalopathy, including autoimmune encephalitis, was negative. Also, our patient improved dramatically after receiving methylprednisolone (a synthetic glucocorticoid, primarily prescribed for its anti-inflammatory and immunosuppressive effects), supporting an immune-mediated mechanism behind his acute presentation. Accordingly, we think the COVID-19 vaccine is the only possible cause of our patient presentation, giving the temporal relationship and the absence of other risk factors for encephalopathy.”
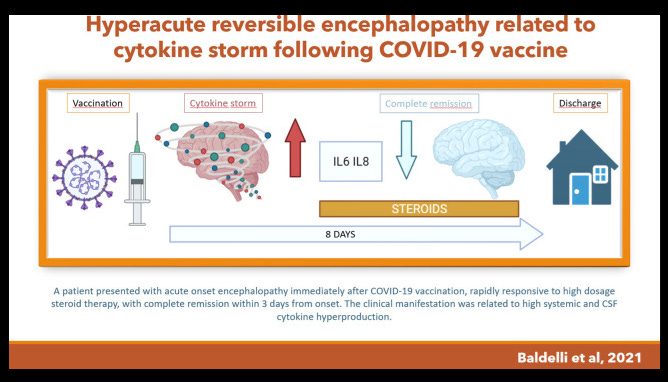
Baldelli6 et al described the first case of hyperacute reversible encephalopathy following COVID-19 vaccination. They reported a patient presented with acute onset encephalopathy, mainly characterized by agitation and confusion, rapidly responsive to high dosage steroid therapy and complete remission within 3 days from onset. The clinical manifestation was related with systemic and CSF (cerebrospinal fluid) cytokine hyperproduction, responsive to steroid therapy. The researchers speculated that the cytokine-storm could be the result of an excessive innate immune response against the vaccine, in a predisposed patient susceptible to autoimmunity.
Zuhorn7 et al reported a case series of three patients with autoimmune encephalitis related to prior ChAdOx1 nCoV-19 vaccination, previously undescribed. Complication of postvaccinal encephalitis after ChAdOx1 nCoV-19 vaccination still appear to be very rare, but need to be diagnosed and treated adequately. The incidence has been estimated to be approximately 8 per 10 million vaccine doses.
Studies have shown the effect of inflammation of the vaccinated people. According to the above Primary Care study, PD symptoms are supposed to be found after less than 2 years (tremor) and 5 to 10 years (epilepsy) of diagnosis. Can the epilepsy & encephalitis cases from the above studies and tremors in the Yellow Card data accelerate the vaccinated to the PD? With no history of neurological complication as studied cases, a person is unlikely to have an accelerated neurodegenerative disease.
https://www.gov.uk/government/publications/coronavirus-covid-19-vaccine-adverse-reactions/coronavirus-vaccine-summary-of-yellow-card-reporting
https://www.medrxiv.org/content/10.1101/2021.09.30.21264336v1
https://www.seizure-journal.com/article/S1059-1311(21)00269-7/fulltext
https://www.cureus.com/articles/56583-two-cases-of-post-moderna-covid-19-vaccine-encephalopathy-associated-with-nonconvulsive-status-epilepticus
https://www.sciencedirect.com/science/article/pii/S2049080121007536
https://pubmed.ncbi.nlm.nih.gov/34284342/
https://onlinelibrary.wiley.com/doi/10.1002/ana.26182